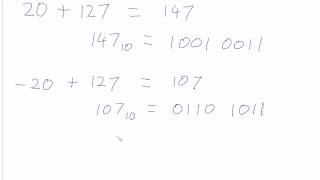Texas Instruments TI84PLUS - Graphing Calculator USB technology : Amazon.de: Stationery & Office Supplies

1 Lecture 5 Floating Point Numbers ITEC 1000 “Introduction to Information Technology” - ppt download

Texas Instruments TI-84 Plus CE Pink Graphing Calculator: Amazon.co.uk: Stationery & Office Supplies
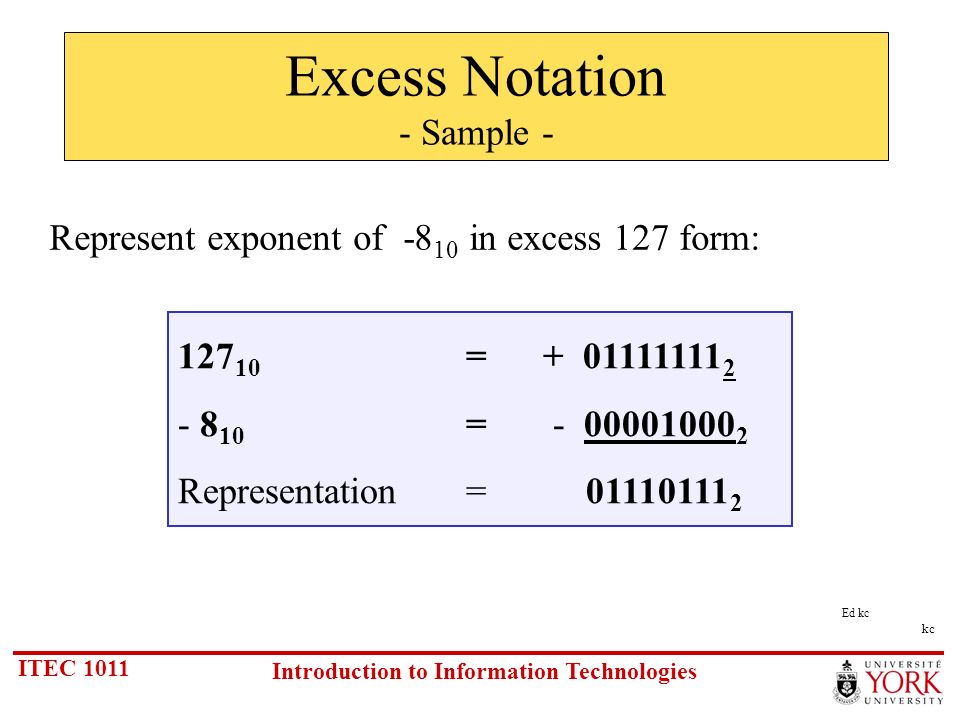
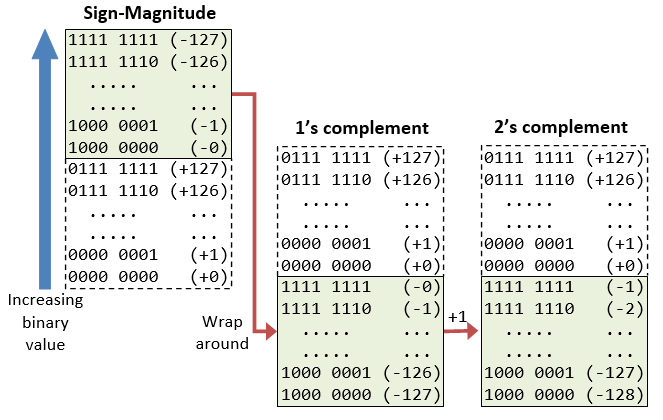
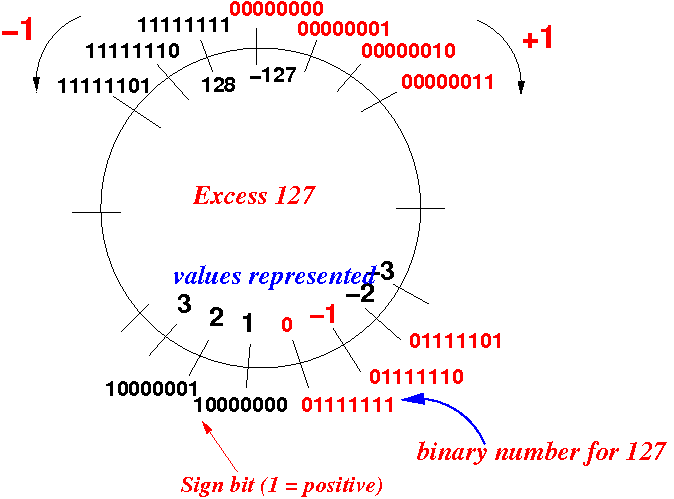
Why do we add 127 to the exponent in IEEE-754 floating number format to get the actual exponent value? - Quora

Risk score calculation flowchart: data inputs, sources, and calculations | Download Scientific Diagram